12.1: Introduction to Cross-Over Designs
- Page ID
- 33195
The simplest cross-over design is a 2-level treatment, 2-period design. If we use A and B to represent the two treatment levels, then we can build the following table to represent their administering sequences.
| Sequence | Period 1 | Period 2 |
|---|---|---|
| 1 | A | B |
| 2 | B | A |
Experimental units are randomly assigned to receive one of the two different sequences. For example, if this were a clinical trial, patients assigned to sequence 2 would be given treatment B first, then after assessment of their condition, given treatment A and their condition re-assessed.
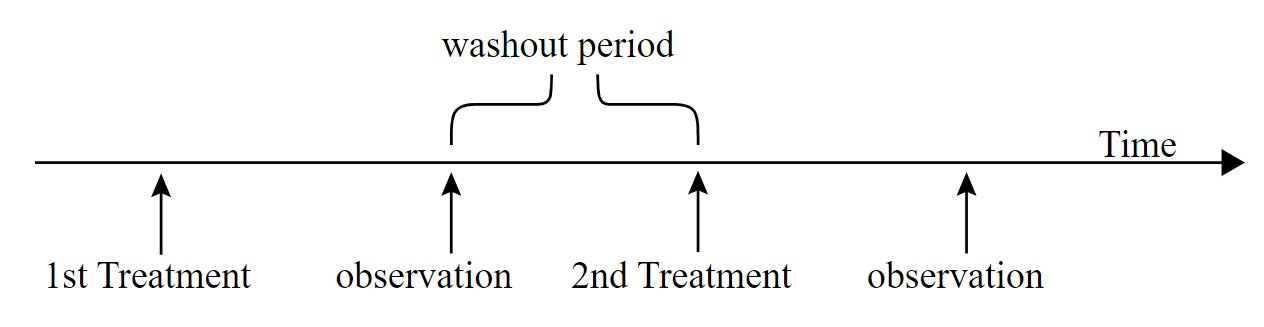
The complicated part of the cross-over design is the potential for carry-over effects. A carry-over effect is when the response to a particular treatment level has been influenced by the previous application of a different treatment level. The presence of carry-over effects is dealt with differently by different researchers in different ways. Having a sufficiently long washout period is one way to reduce carry-over effects. A washout period is a gap in time between the application of the treatment levels such that any residual effect of a previous treatment level has been dissipated and there is no detectable carry-over effect.
.png?revision=1&size=bestfit&width=550&height=142)
However, there may be instances where significant carry-over effects may exist and sufficiently long washout periods may not be practically feasible. In such situations, an adjustment for carry-over effects would be appropriate during the statistical analysis.
If the treatment has only 2 levels, it is sufficient to simply include a "sequence" categorical variable in the model to assess the presence of a carry-over effect. If the sequence variable is significant, then a detectable carry-over effect exists.
With more than two treatment levels, the complexity of the analysis rises sharply. For 3 levels of treatment, 3 periods will be needed, and now we have 3! = 6 sequences to consider. What is needed in this case, in addition to a sequence variable, is a way to adjust the assessment of treatment effects for the presence of carry-over effects. This can be accomplished with a set of coded covariates in a repeated-measures ANCOVA.


